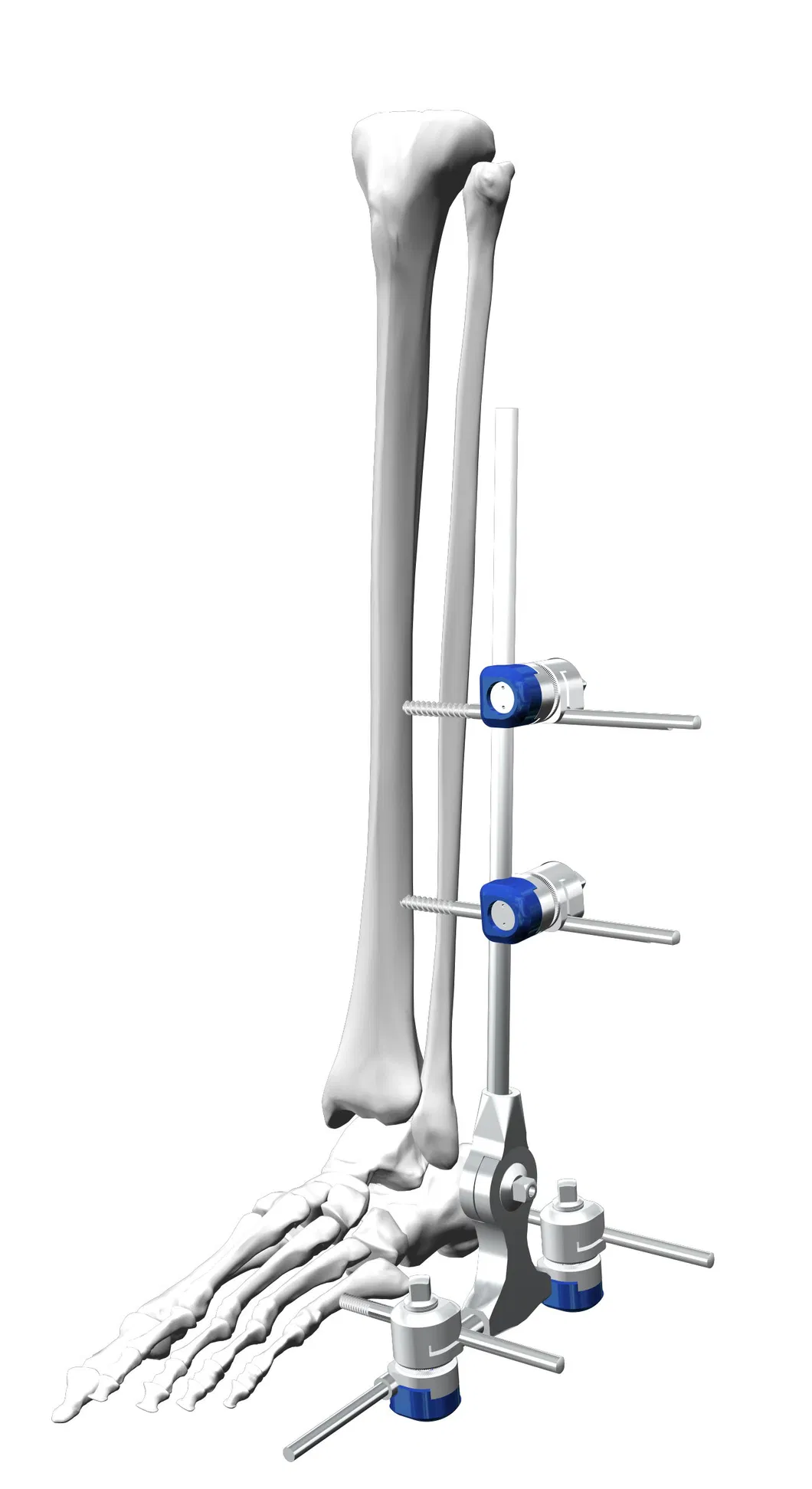
Frequently Asked Questions
Q1: What materials are used in the TY-LP-383 External Fixator?
A1: The device is constructed using high-quality Carbon Fiber and Stainless Steel for optimal strength and durability.
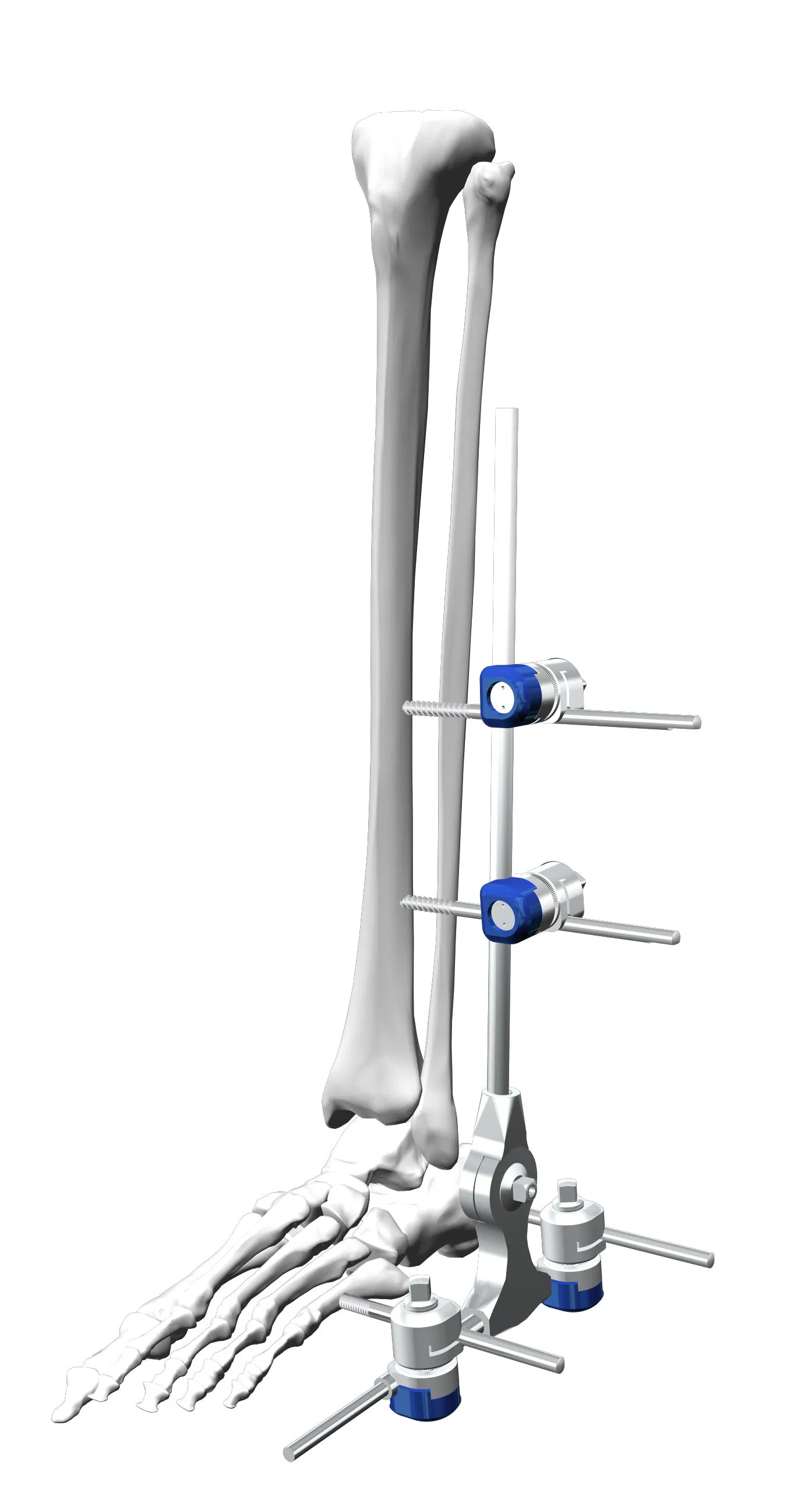
Q2: What are the primary medical applications for this device?
A2: It is specifically designed for medical orthopedic surgical procedures, including fracture fixation and the correction of deformities in the ankle.
Q3: Do you provide OEM services for this product?
A3: Yes, OEM services are available to meet specific customization requirements.
Q4: What certifications does the ankle external fixator hold?
A4: The product is fully certified with CE and ISO13485 standards, ensuring compliance with international medical quality regulations.
Q5: What is the warranty and after-sales support policy?
A5: We offer a one-year warranty and provide ongoing online technical support for all our surgical instruments.
Q6: What is included in the standard Dissection IV package?
A6: The standard set includes one ankle fixator, four pin-rod couplings, and four one-step pins of varying sizes (Φ5×150 and Φ5×120).
 MGMT Orthopedic Implants
MGMT Orthopedic Implants