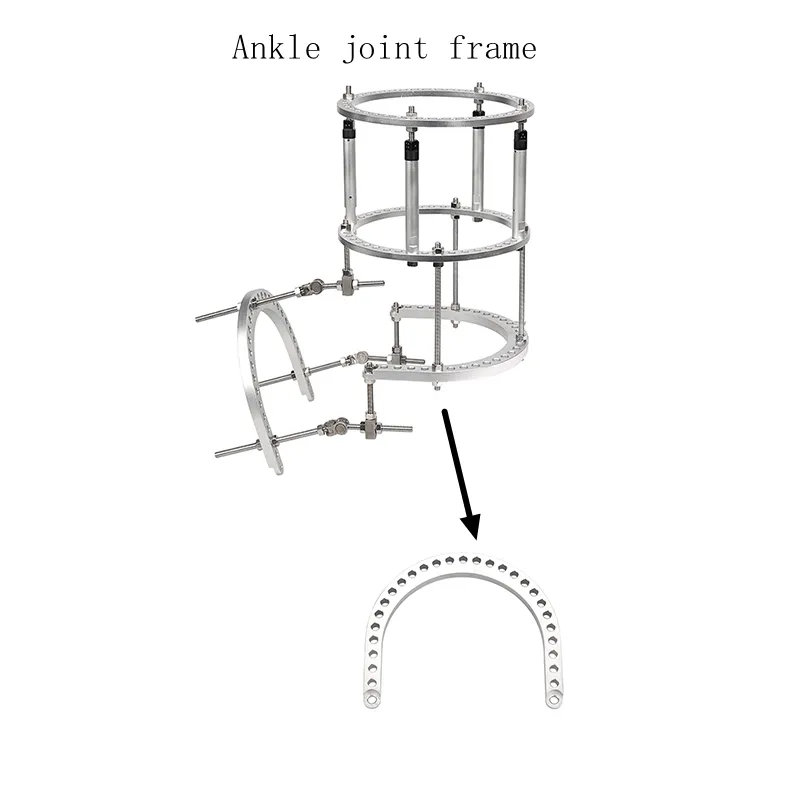
Q1: What are the primary applications for the HOTL03 U-ring fixator?
A: It is primarily used for bone fracture fixation and various types of deformity correction in adults, particularly for ankle joint framing.
Q2: Which materials are available for this external fixator?
A: The fixator is available in high-grade Aluminum and Stainless Steel (Ss) to ensure durability and biocompatibility.
Q3: Does the device require a second surgery for removal?
A: No, one of its main advantages is that it can be removed in a clinical setting, making a second major surgery unnecessary.
Q4: What sizes of U-rings are available?
A: We offer multiple sizes including φ120, φ140, φ150, φ160, φ180, and φ200 to accommodate different patient requirements.
Q5: Is the system compatible with minimally invasive surgery?
A: Yes, the design supports minimally invasive procedures, which helps preserve the blood supply to the bone and promotes faster healing.
Q6: What certifications do your medical products hold?
A: Our products are fully certified with CE and ISO13485 standards, ensuring compliance with international medical quality requirements.

 MGMT Orthopedic Implants
MGMT Orthopedic Implants