❓ Frequently Asked Questions
Q: What materials are used for the suture anchors?
A: The anchors are manufactured from Ti6Al4V alloy, which strictly complies with ISO5832-3:2016 standards for surgical implants.
Q: Is the suture anchor system provided sterile?
A: Yes, all systems are EO (Ethylene Oxide) sterilized and packaged in medical-grade blister boxes to ensure safety for orthopedic surgery.
Q: Do you offer OEM or customization services?
A: Yes, we provide flexible OEM services, including custom design and packaging solutions tailored to specific brand requirements.
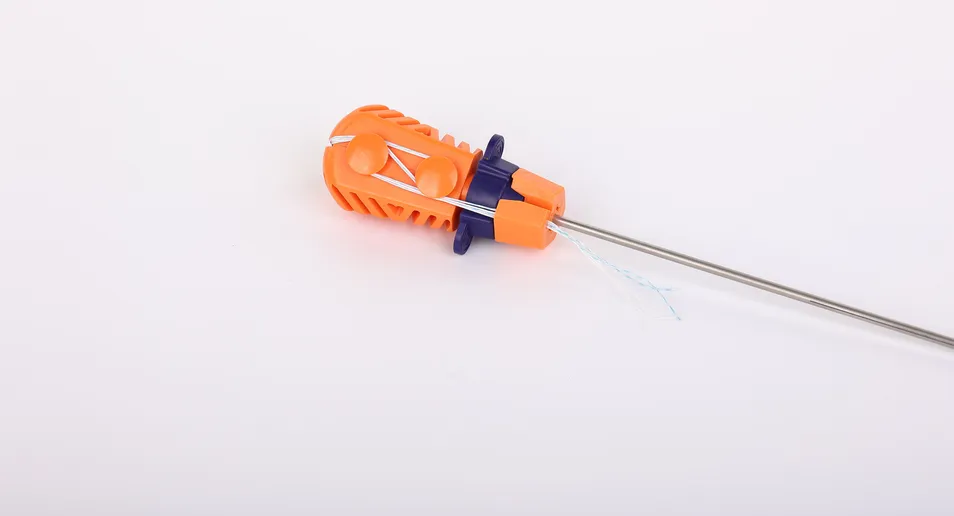
Q: What are the primary applications for this system?
A: This system is specifically designed for orthopedic surgeries to securely connect soft tissue to bone using high-strength sutures and anchors.
Q: What certifications do your medical instruments hold?
A: Our products are fully compliant with international quality benchmarks, holding both CE certification and ISO 13485 standards.
Q: Can I request samples for evaluation?
A: Yes, we provide samples for quality evaluation. Please reach out to discuss the specific sizes and lead times required.




 MGMT Orthopedic Implants
MGMT Orthopedic Implants